In a collaborative project, the UO’s Patrick Phillips tackles a problem of reproducibility while studying potential anti-aging compounds
Worms. Might they help us live a healthier and longer life?
Extending human life in ways that keep people both healthy and productive is a goal of many scientists, including the UO’s Patrick Phillips.
His latest project, which he leads in collaboration with two other U.S. institutions, may not immediately move us closer to extending human life beyond the national average of 79. It has, however, opened a window on how basic research — that which seeks fundamental knowledge about how something works — should be done to harness robust results that speed progress toward medical advances.
In a new paper published Feb. 21 in the high-profile journal Nature Communications, Phillips and 33 collaborators got right to the heart of the challenge: Too many laboratory findings are not reproducible, and the genetic makeup of model organisms often responds differently to compounds thought to offer promise.
Overcoming the hurdles
“Aging is universal. It is complex. Individuals die for many different reasons, so there is a lot of noise in the system,” said Phillips, a professor of biology and acting executive director of the Phil and Penny Knight Campus for Accelerating Scientific Impact. “It is a challenge to figure out the elements necessary to change the process. To do this you have to approach the question at a scale that has never been done before. That’s what our paper is about.”
In their study, Phillips’ nine-member UO team and researchers from the Buck Institute for Research on Aging in California and Rutgers University in New Jersey carefully carried out experiments using identical protocols. They simultaneously tested the effects of 10 different compounds on life extension across 22 diverse genetic backgrounds drawn from three species of roundworms.
“This is the largest aging study that has ever been done on an animal — hundreds of thousands of individuals have been tested,” Phillips said.
“Our study indicates that even when following the same methods, insufficient replication of trials could account for failures to reproduce previous studies,” the research team noted in the paper. “Our focus on rigorously adhering to defined methods to reduce variability between sites necessitated making choices about specific methodologies for which there was no standard across the field.”
Locations of worm strains
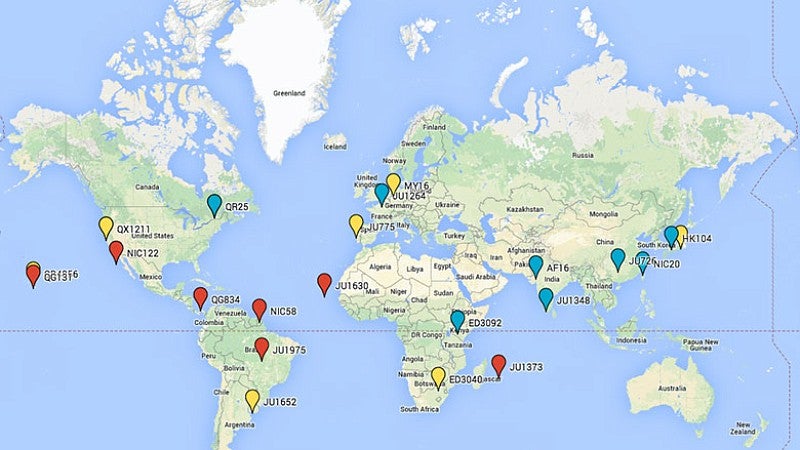
Across the labs, the researchers identified six compounds that extended the lifespan in one strain of worms. Overall, two compounds had positive results across the various strains, with an amyloid dye, Thioflavin T, being the most effective; two other compounds offered promise under specific conditions. Genetic differences among the species are comparable to those found in mice and humans, the researchers noted.
More details about the science and Thioflavin T are covered in a news release issued by the Buck Institute.
Future experiments, Phillips said, will test these and other promising compounds in genetically diverse strains of roundworm species to see how they perform. Eventually, the most widely acting compounds could advance into testing in other animal models and, eventually, in human clinical trials.
The research emerged from three-year grants to each of the three collaborating institutions from the National Institutes of Health. It is part of an extension of the National Institute on Aging’s decade-old Intervention Testing Program that has targeted aging studies using mice at three other institutions. The roundworm project is known as the Caenorhabditis Intervention Testing Program.
Roundworms, which have a lifespan of two to three weeks, have a simple genetic makeup that is similar to mice, which in laboratories can live up to three years. Thus, Phillips noted, more individual worms can be used more cheaply in the course of experiments that span the life cycle.
Compounds that have been found to extend life in worms and mice have proved so far to be limited to organisms with a particular genetic background.
Roundworms
“This is a dark side of studying a model organism,” Phillips said. “You have genetic uniformity in worms and mice, but humans are not genetically uniform. We know that different individuals respond differently to drugs and that the cause of disease is often different in each individual. Overcoming those limitations is a big part of the push toward personalized medicine.”
From the outset, he said, the roundworm project has been about reproducibility in a way that mirrors the approaches used by the institutions studying mice.
“We’ve had to invest a lot of time in coordinating activities,” Phillips said. “That’s often an unstated part of the difficulty of doing science. For this, we’ve written hundreds of pages of standard operating procedures to try to normalize the research process.
“There is a history in aging studies where one lab finds a result but another lab cannot reproduce it,” he said. “Cancer studies are the same. Only about 25 percent of studies can be reproduced with similar results. This is a big emerging issue in science now, so we feel like our study is one of the best on reproducibility that has ever been produced.”
For the project, the leaders of the three labs brought different specialties of nematode biology to the table: Phillips is an expert in evolutionary genetics; Gordon J. Lithgow of the Buck Institute is a specialist on chemical interventions; and Rutgers’ Monica Driscoll is an aging and health expert.
Can we expect to see extended human lifespans soon?
“What we find in this worm may or may not work in mice or humans,” Phillips said. “We’re looking at things that affect fundamental cellular processes that are conserved genetically across all animals.”
Promise of the Knight Campus
Carrying basic research forward is a goal of the Knight Campus, a $1 billion initiative designed to accelerate the cycle of generating impact from discoveries. The Knight Campus, which has seen some recent behind-the-scenes progress on staffing and the selection of architects and general contractors, will foster exchanges of ideas among basic-science researchers with applied scientists and entrepreneurs to foster that translational process.
“With this research, you are seeing the classic impact cycle,” Phillips said. “You have a guy working in a most esoteric part of evolutionary biology — something that you’d generally think could have no general impacts — just to gain understanding about something about the world. It is important, but in terms of affecting human health, who knows? Understanding genetic variation is being recognized as being more important each day. And so what once seemed esoteric is now important for understanding translational medicine.
“As scientists expand into studying stress and aging in terms of natural genetic variation in different species, then my area’s unique contributions fit into a broader scale. We’re looking at compounds in a way that’s never been done before. We are identifying compounds that can affect health and aging,” he said. “What do we do with that?
“The point is not to make worms live a long time. It’s how we use the information. How might this translate a decade from now into something that could go into human clinical trials to try to help people to live longer healthier lives? Can we turn this basic research into something that is relevant? Are there potential drugs that could?
“That could be a Knight Campus story,” Phillips said.
—By Jim Barlow, University Communications




